Home / Blog
Blog Categories
Recent Posts
Tags
- API Impurity
- API Manufacturing
- Career
- Characterization
- Chemist
- custom synthesis
- Degradation Impurities
- Drug Development
- Drug Product
- drug safety
- Drug Substance
- Excipients
- Genotoxic Impurities
- Green Chemistry
- HPLC
- HPLC Method
- Impurities
- Impurity
- impurity profiling
- Internal Reference Standards
- Job
- LC-MS
- M.Sc. Organic Chemistry
- Mass Spectrometry
- MS/MS
- NDSRI
- nitrosamine
- nitrosamine impurity
- Pharmaceutical Analysis
- Process Impurities
- Reference Standards
- Research Chemist
- Working Standards
Welcome to SynThink Blog
Here, we keep you posted about the latest updates and insights into pharmaceutical impurities. We understand that impurities in pharmaceuticals can significantly impact their safety and efficacy, and it is crucial to maintain stringent standards to ensure quality of the products. Our blog site provides the most accurate and up-to-date information on impurity standards, regulations, and guidelines in the pharmaceutical industry. We hope you find our blogs informative and valuable.

Understanding ICH Q3C: Residual Solvents in APIs Explained
The International Council for Harmonisation (ICH) provides clear guidelines for the solvents’ impurities in Active Pharmaceutical Ingredients (APIs).

2026 Update on Pharmaceutical Standards
For multinational pharmaceutical companies, CDMOs, biotech innovators, and API manufacturers operating across multiple jurisdictions, regulatory alignment is now a strategic priority

ICH Q3A vs Q3B: API and Drug Product Impurity Guidelines Explained
This comprehensive guide explains how industry professionals can benefit from the ICH Q3A and Q3B guidelines for API and drug product impurities.

FDA Nitrosamine Categorization: Small-Molecule Nitrosamine and NDSRIs
The issue of nitrosamine impurities has become one of the most significant regulatory concerns in the pharmaceutical industry since 2018.

Green Chemistry Approaches in API Impurity Synthesis
Green chemistry is no longer optional in pharmaceutical research. It is essential because the regulatory agencies expect safer processes for chemical development. Also, the companies demand cost efficiency and cleaner reactions.
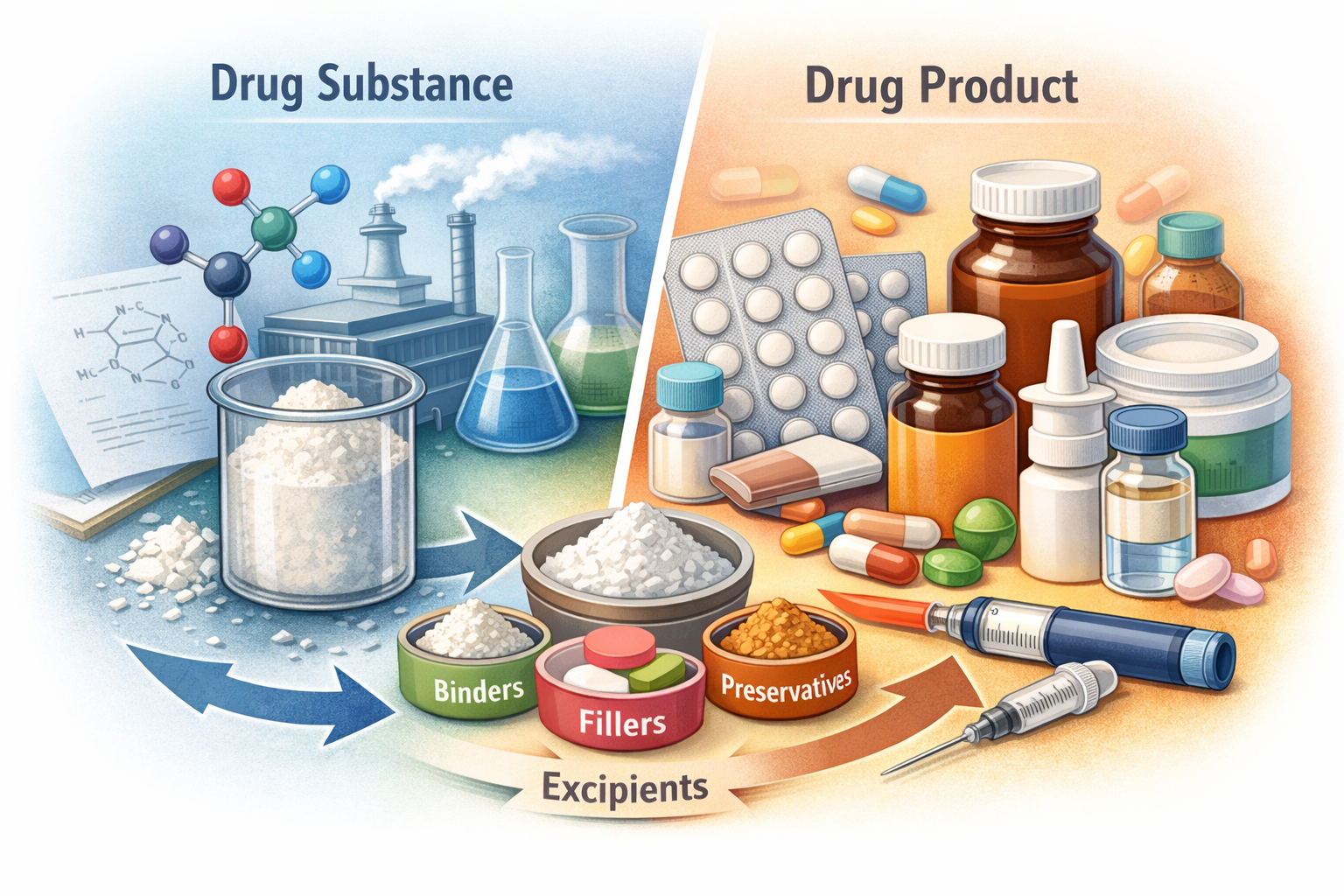
Drug Substance vs Drug Product: A Simple, Clear Guide for Beginners
If you work in pharmaceuticals, chemistry, biotech, or regulatory affairs, you will hear two terms again and again: drug substance and drug product.
They sound similar, but they mean very different things.

Difference between Process-Related and Degradation-Related Impurities
Understanding the distinctions between Process Impurities and Degradation Impurities is crucial in pharmaceutical development and quality assurance…

Structure Elucidation of Unknown Impurity using LC-MS/MS
Impurity analysis in pharmaceuticals is a critical quality control measure. Impurities, arising during synthesis, storage, or handling, can significantly affect a drug’s safety, efficacy, and stability…

Advancements in LC-MS Technology
Liquid Chromatography-Mass Spectrometry (LC-MS or LC/MS or LCMS) is an indispensable technology in the realm of pharmaceutical analysis…

The Nitrosamine puzzle: What We Learned in the Last Five Years
The Nitrosamine (NA) issue, often termed the “Saga,” has been a focal point of attention for both the pharmaceutical sector and regulatory entities over the past half-decade…

Impurity Profiling
Impurity profiling plays a crucial role in pharmaceutical product development and quality control. It involves the identification, quantification, and characterization of impurities present in drug substances and drug products…

SynThink: Empowering Fresh M.Sc. Organic Chemistry Graduates for a Future of Scientific Excellence
Welcome, fresh M.Sc. Organic Chemistry graduates! Choosing the right path is crucial as you step into the professional world…
